  
The basic requirements of our life-shelter, food, clothes, medicines are basically chemical compounds. Atomic absorption spectroscopy is based on absorption of light by free metallic ions. 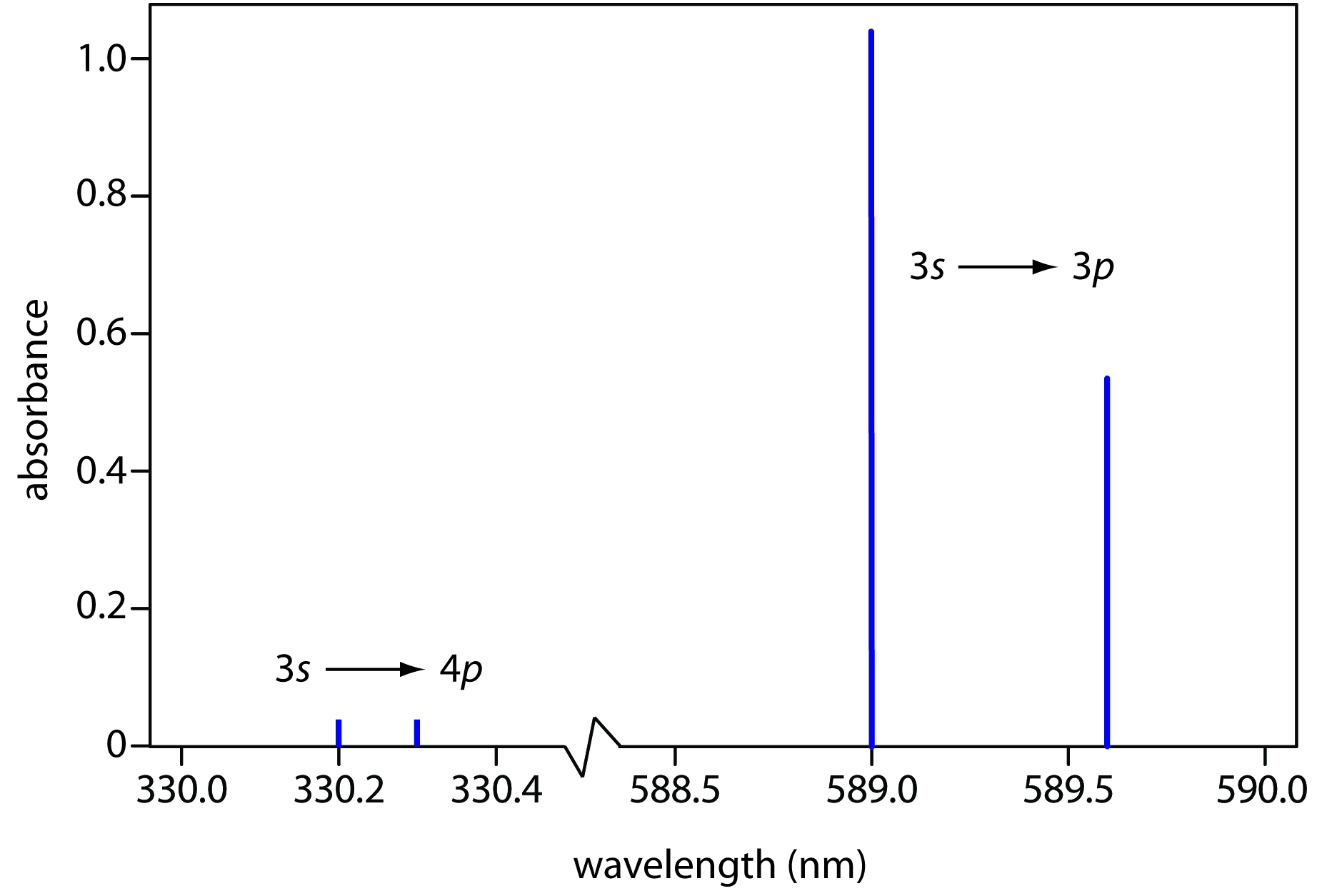
The observation of fine structure revealed that an orbital energy level diagram does not completely describe the energy levels of atoms. Atomic spectroscopy is an excellent analytical tool used for the detection and measurement of elements in a sample with high precision and confidence. The Atomic spectra are defined as the Spectrum of frequencies of electromagnetic radiation emitted or absorbed during transitions of Electrons between. Atomic Spectra Atomic Spectra: Concept with Definition, Spectral Series, Bohr’s Atom & Rydberg Formula Last updated on Download as PDF Overview Test Series Chemistry plays an important role in everyday life. Atomic absorption spectroscopy ( AAS) and atomic emission spectroscopy ( AES) is a spectroanalytical procedure for the quantitative determination of chemical elements by free atoms in the gaseous state. An atom consists of a number of negatively charged electrons bound to a nucleus containing an equal number of positively charged protons. \): When light from a hydrogen gas discharge tube is passed through a prism, the light is split into four visible lines.\) line in the Balmer series at 656.279 nm was understood as resulting from a single transition of an electron from the n = 3 energy level to the n = 2 level. The range of characteristic frequencies of electromagnetic radiation that are readily absorbed and emitted by an atom. major reference In spectroscopy: Basic atomic structure The emission and absorption spectra of the elements depend on the electronic structure of the atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
